Black Root Rot of Holly
ID
450-606 (SPES-569P)
EXPERT REVIEWED
Introduction
Black root rot is a fungal root disease that is a serious and extremely common problem on Japanese holly (Ilex crenata), a commonly used evergreen landscape shrub. Inkberry holly (Ilex glabra), blue or Meserve holly (Ilex crenata) are also very susceptible to black root. The disease is not as commonly diagnosed on blue holly and inkberry holly as on Japanese holly in the Virginia Tech Plant Disease Clinic. The black root rot pathogen is soil-borne and can be introduced into a landscape on infected nursery plants. Chinese holly (Ilex cornuta) and English holly (Ilex aquifolium), are resistant to the black root rot pathogen.
Symptoms
Black root rot is named for the black lesions that commonly occur on infected roots. Symptoms on holly include foliar yellowing and generally off-color foliage (e.g. lighter green than normal), leaf drop, stunting and sectional dieback, eventually progressing to plant death (figure 1). Young holly plants in the nursery can be killed within weeks as a result of severe root destruction by the fungus; however, mature plants decline more slowly.

The Black Root Rot Pathogen
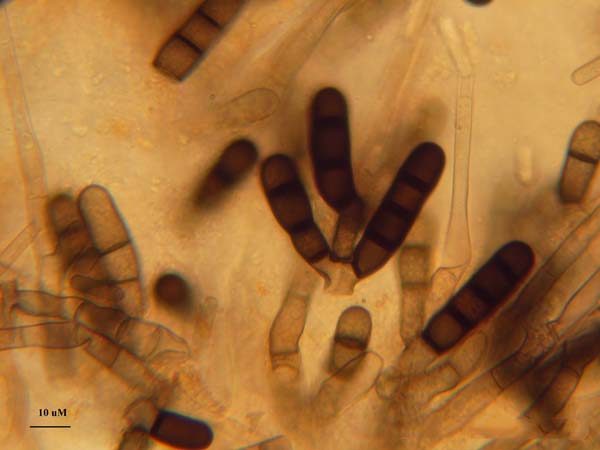
The black root rot pathogen, Berkeleyomyces basicola (synonym, Thielaviopsis basicola), is a soilborne fungal pathogen that produces long-lived survival structures, chlamydospores, which survive in the soil for long periods of time, even in the absence of a host plant (figure 2). The chlamydospores are darkly pigmented and when present in root tissue, cause the roots to become very dark, thus the name of the disease: black root rot. The pathogen is ubiquitous in many soil environments, but can also be introduced into new locations on black root rot-infected plants material (figure 3). For home growers, avoidance of introduction of infected plant material is a good approach to minimize the chance of this serious root disease problem in the home landscape.

Host Range
Black root rot is also a serious problem on many herbaceous ornamentals, such as pansy, viola, vinca, petunia, salvia, calibrachoa, coral bells, poinsettia and phlox, and several important vegetable and field crops, including bean, tobacco, and peanut. However, black root rot commonly affects only a few species of woody ornamentals grown in Virginia landscapes. These include Japanese holly, inkberry (I. glabra), and blue or Meserve holly (I. x meserveae). Yaupon holly (I. vomitoria) and American holly (I. opaca) are moderately resistant to the disease, whereas English holly (I. aquifolium) and Chinese holly (I. cornuta) are highly resistant.
Avoidance and Management
Landscape plants:
- Avoid locations with overly wet soil conditions that predispose plants to root disease or correct poorly draining soils prior to planting.
- When purchasing holly for new plantings.
- Avoid the use of Japanese holly, since it is so commonly afflicted by black root rot.
- Avoid purchasing any holly that is off-color or has browning foliage or dieback.
- Consider installing black root rot-resistant holly OR other woody ornamental shrub genera (other than Ilex species).
- In established plantings, when Japanese holly or other black root rot-susceptible holly are diagnosed with black root rot: If the plant(s) are already show foliar symptoms, significant root loss has already occurred and, therefore, plants will not benefit from fungicide treatment. Such plants should be removed.
- Preventative fungicides can be used to treat hollies not showing symptoms of black root rot, but fungicide treatment will need to be ongoing. Therefore, in a landscape situation, black root rot-resistant holly or other genera of woody shrubs should be considered for replacement plants. Most genera of woody shrubs (i.e. those other than black root rot-susceptible Ilex species) can be grown successfully in soil infested with the black root rot fungus, as can black root rot-resistant holly species, such as Chinese holly or English holly (Note that English holly is hardy only in zones 7-9, so would not be appropriate for all locations in Virginia.).
- Refer to the Home Grounds and Animals Pest Management Guide (VCE Publication 456-018) https://www.pubs.ext.vt.edu/456/456-018/456-018.html for fungicide recommendations for black root rot, but keep in mind that fungicides are not curative and the fungus will persist in the soil for a long time.
Greenhouse/Nursery plants:
Strict sanitation is key to avoiding black root rot, since it can be introduced to the nursery or greenhouse and spread via infested tools, pots, flats, benches, equipment, soil, soilless potting mix and some insect pests.
- Fungus gnats and shore flies can spread the fungal pathogen, so manage these pests.
- For propagation use only healthy mother plant material and practice strict sanitation during propagation by disinfesting surfaces and tools and using new or properly cleaned and sanitized flats, pots. Use soilless media and never re-use potting media.
- Maintain potting medium pH at 5.5, since the black root rot pathogen is favored by neutral to alkaline soil conditions (pH 6 to 7).
- If re-using containers or flats, they must be thoroughly power-washed free of all debris and surface disinfested with a commercial disinfestant or 10% bleach solution prior to use.
- Avoid cool potting media temperatures, overly wet media conditions and, excessive fertilization.
- If plants are diagnosed with black root rot, remove plants from the nursery/greenhouse, along with associated potting media, flats and pots. Disinfect surfaces with a commercial disinfectant or 10% household bleach after surfaces are washed free of debris.
- Fungicides can be used preventively, but are not effective on plants already showing symptoms of black root rot. Refer to the Horticultural and Forest Crops Pest Management Guide (VCE Publication 456-017, https://www.pubs.ext.vt.edu/456/456-017/456-017.html) for fungicide information.
Diagnosing the Disease
The Virginia Tech Plant Disease Clinic can diagnose this disease and other plant diseases. Refer to the Plant Disease Clinic website (https://bit.ly/VTplantclinic)for the current diagnostic form, fees, and instructions on collecting an appropriate diagnostic sample and submitting samples to the Plant Disease Clinic.
References
Chase, A.R. and Daughtrey, M. April 2013. Combating Black Root Rot. Greenhouse Product News. https://gpnmag.com/article/combating-black-root-rot/
Wills, W. H. and R.C. Lambe. 1978. Pathogenicity of Thielaviopsis basicola to Japanese holly (Ilex crenata). Plant Disease Reporter 62(10):859-863. Diseases of Woody Ornamentals and Trees in Nurseries. 2001. Edited by R.K. Jones and D. M Benson. APS Press, The American Phytopathological Society.
Wills, W. H. and R.C. Lambe. 1978. Pathogenicity of Thielaviopsis basicola to Japanese holly (Ilex crenata). Plant Disease Reporter 62(10):859-863.
Wills, W. H. and R.C. Lambe. 1978. Pathogenicity of Thielaviopsis basicola from Japanese holly (Ilex crenata)to some other host plants. Plant Disease Reporter 62(12):1102-1106.
Scan to visit the Plant Disease Clinic website

Virginia Cooperative Extension materials are available for public use, reprint, or citation without further permission, provided the use includes credit to the author and to Virginia Cooperative Extension, Virginia Tech, and Virginia State University.
Virginia Cooperative Extension is a partnership of Virginia Tech, Virginia State University, the U.S. Department of Agriculture (USDA), and local governments, and is an equal opportunity employer. For the full non-discrimination statement, please visit ext.vt.edu/accessibility.
Publication Date
February 7, 2024



